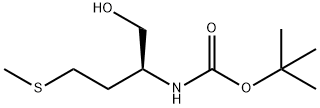
BOC-MET-OL synthesis
- Product Name:BOC-MET-OL
- CAS Number:51372-93-1
- Molecular formula:C10H21NO3S
- Molecular Weight:235.34
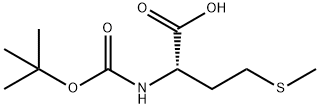
2488-15-5

51372-93-1
General procedure for the synthesis of (S)-2-(tert-butoxycarbonylamino)-4-methylthio-1-butanol from Boc-L-methionine: Boc-L-methionine (49.8 g, 0.5 mol) and 250 mL of tetrahydrofuran (THF) were added to a 500 mL three-neck flask. The reaction mixture was cooled to 0-5 °C in an ice-salt bath with stirring. Sodium borohydride (19 g, 0.5 mol) was slowly added, followed by 50 mL of methanol. Next, 100 mL of tetrahydrofuran solution of iodine (127 g, 0.5 mol) was added dropwise. The reaction system was heated to reflux and the reaction was continuously stirred for 2 hours. The progress of the reaction was monitored by thin layer chromatography (TLC) and after confirming the complete consumption of raw materials, the reaction mixture was cooled in an ice water bath. Saturated ammonium chloride solution was added to quench the reaction. After approximately 100 mL of THF was removed by evaporation using a reduced pressure evaporator, the residue was extracted with ethyl acetate (300 mL). The organic layer was washed sequentially with dilute hydrochloric acid, saturated sodium bicarbonate solution and saturated sodium chloride solution. After drying with anhydrous sodium sulfate, the crude product (120 g) was concentrated to give an oil. The crude product was purified by column chromatography to give the purified product (97.5 g) in 85% yield. The structure of the product was confirmed by 1H NMR (400 MHz, CDCl3) and mass spectrometry (MS): 1H NMR δ 11.64 (br, 1H), 6.85 (br, 1H), 4.55-4.48 (m, 1H), 2.53 (t, J = 4.9 Hz, 2H), 3.44 (s, 3H), 2.05 (s, 3H), 2.02- 1.87 (m, 2H), 1.48 (s, 9H); MS (M + + 1): 236.
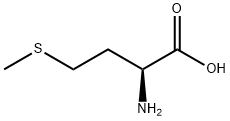
63-68-3
953 suppliers
$5.00/25g

51372-93-1
108 suppliers
$11.00/1g
Yield:-
Steps:
Multi-step reaction with 3 steps
1: aq. NaOH / dioxane / 20 °C
2: NEt3 / tetrahydrofuran / -10 °C
3: 74 percent / NaBH4 / H2O / 0 °C
References:
Catalano, John G;Deaton, David N;Furfine, Eric S;Hassell, Annie M;McFadyen, Robert B;Miller, Aaron B;Miller, Larry R;Shewchuk, Lisa M;Willard Jr., Derril H;Wright, Lois L [Bioorganic and medicinal chemistry letters,2004,vol. 14,# 1,p. 275 - 278]

2488-15-5
284 suppliers
$10.00/1g

51372-93-1
108 suppliers
$11.00/1g
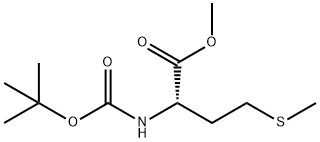
33900-24-2
3 suppliers
inquiry

51372-93-1
108 suppliers
$11.00/1g
24424-99-5
865 suppliers
$13.50/25G

2899-37-8
224 suppliers
$22.00/1g

51372-93-1
108 suppliers
$11.00/1g
![Ethanamine, N-[(3,5-dichlorophenyl)methylene]-2,2-diethoxy-](/CAS/20210305/GIF/1000210-73-0.gif)
1000210-73-0
0 suppliers
inquiry

51372-93-1
108 suppliers
$11.00/1g